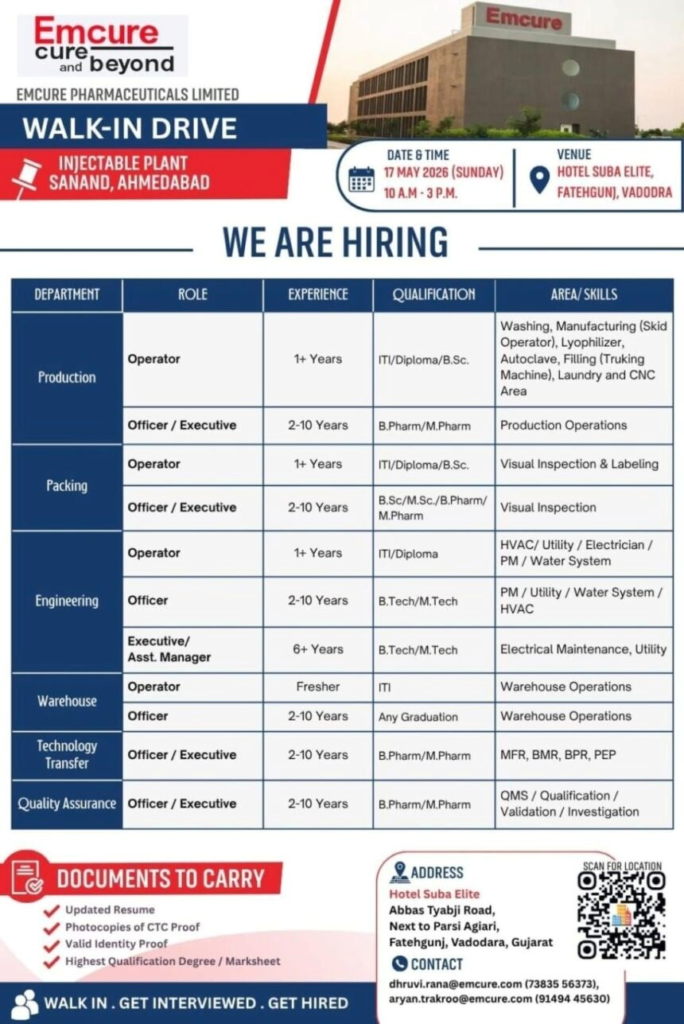
Emcure Pharmaceuticals is conducting a Walk-In Drive for multiple positions at its Injectable Plant in Sanand, Ahmedabad. This is a great opportunity for pharma jobs seekers in Production, Packing, Quality Assurance, Engineering, Warehouse, and Technology Transfer. Join a leading pharmaceutical company for exciting pharmaceutical careers in India.
Contents
About the Company
Emcure Pharmaceuticals Limited is a leading Indian multinational pharmaceutical company headquartered in Pune. Established in 1981, Emcure has grown into a trusted global player with a strong presence in over 70 countries.
The company is known for its innovation, high-quality manufacturing standards, and regulatory excellence, including USFDA-approved facilities. With a focus on complex injectables, oncology, and specialty formulations, Emcure continues to deliver affordable and effective healthcare solutions worldwide while maintaining strong emphasis on quality, compliance, and employee development.
Job Details
- Company Name: Emcure Pharmaceuticals Limited
- Experience: 1–10+ years (varies by role)
- Qualification: ITI, Diploma, B.Sc., B.Pharm, M.Pharm, B.Tech, M.Tech (varies by role)
- Location: Sanand, Ahmedabad, Gujarat (Injectable Plant)
- Work Type: On-site
Job Description
Emcure Pharmaceuticals Limited is hiring talented professionals for its state-of-the-art Injectable manufacturing facility at Sanand, Ahmedabad. This walk-in drive offers multiple openings across Production, Packing, Engineering, Warehouse, Technology Transfer, and Quality Assurance departments for both Operator and Officer/Executive levels.
- Production – Operator Department: Production
- Market: Injectable Manufacturing
- Role: Injectable production operations
- Experience: 1+ years
- Education/Qualification: ITI/Diploma/B.Sc.
- Production – Officer/Executive Department: Production
- Market: Injectable Manufacturing
- Role: Production operations management
- Experience: 2–10 years
- Education/Qualification: B.Pharm/M.Pharm
- Packing – Operator Department: Packing
- Market: Injectable Manufacturing
- Role: Visual inspection and labeling
- Experience: 1+ years
- Education/Qualification: ITI/Diploma/B.Sc.
- Packing – Officer/Executive Department: Packing
- Market: Injectable Manufacturing
- Role: Visual inspection operations
- Experience: 2–10 years
- Education/Qualification: B.Sc/M.Sc/B.Pharm/M.Pharm
- Engineering – Operator Department: Engineering
- Market: Pharmaceutical Manufacturing
- Role: HVAC, Utility & Maintenance
- Experience: 1+ years
- Education/Qualification: ITI/Diploma
- Engineering – Officer/Executive/Asst. Manager Department: Engineering
- Market: Pharmaceutical Manufacturing
- Role: PM, Utility, Water System, HVAC
- Experience: 2–10 years
- Education/Qualification: B.Tech/M.Tech
- Warehouse – Operator Department: Warehouse
- Market: Pharmaceutical Manufacturing
- Role: Warehouse operations
- Experience: 6+ years (Electrical/Utility preferred)
- Education/Qualification: ITI/Any Graduation
- Warehouse – Officer Department: Warehouse
- Market: Pharmaceutical Manufacturing
- Role: Warehouse operations
- Experience: 2–10 years
- Education/Qualification: Any Graduation
- Technology Transfer – Officer/Executive Department: Technology Transfer
- Market: Injectable Manufacturing
- Role: Technology transfer activities
- Experience: 2–10 years
- Education/Qualification: B.Pharm/M.Pharm
- Quality Assurance – Officer/Executive Department: Quality Assurance
- Market: Pharmaceutical Manufacturing
- Role: QMS, Validation & Investigation
- Experience: 2–10 years
- Education/Qualification: B.Pharm/M.Pharm
Skills/Qualifications
- Hands-on experience in injectable manufacturing (washing, filling, lyophilizer, autoclave)
- Strong knowledge of GMP, USFDA, and regulatory compliance
- Equipment operation and maintenance skills (HVAC, Utility, Water System)
- Experience in visual inspection and packing operations
- Warehouse management in regulated environment
- QMS, validation, qualification, and investigation expertise
- Teamwork and safety consciousness
Key Responsibilities
- Operate injectable manufacturing equipment
- Perform visual inspection and labeling
- Execute preventive maintenance activities
- Manage warehouse operations efficiently
- Handle technology transfer documentation
- Ensure QMS, validation, and compliance
- Support production and packing targets
Benefits/Perks
- Career growth in a USFDA-approved injectable facility
- Learning opportunities with advanced technology
- Competitive salary and benefits package
- Professional and supportive work culture
- Global exposure in a multinational pharma leader
How to Apply
Attend the Walk-In Interview with all required documents. Alternatively, email your updated CV to: dhruvi.rana@emcure.com or aryan.trakroo@emcure.com
For more pharma jobs and opportunities, visit Pharma Recruiter.
Walk-in Interview Details
- Date: 17 May 2026 (Sunday)
- Time: 10:00 AM to 3:00 PM
- Venue: Hotel Suba Elite, Abbas Tyabji Road, Next to Parsi Agiari, Fatehgunj, Vadodara, Gujarat
- Contact/Email: dhruvi.rana@emcure.com (73835 56373) / aryan.trakroo@emcure.com (91494 45630)
Why You Should Join
Emcure Pharmaceuticals fosters a culture of excellence, innovation, and care. Join a recognized industry leader with strong regulatory compliance and rapid growth. Enjoy long-term career stability, continuous learning, and the opportunity to contribute to high-quality injectable medicines that impact patients globally.
FAQs
Q1: What is the minimum experience required?
A: Experience starts from 1+ year for Operator roles and 2–10 years for Officer/Executive positions.
Q2: Which qualifications are eligible?
A: ITI, Diploma, B.Sc., B.Pharm, M.Pharm, B.Tech, and M.Tech as per the specific role.
Q3: When and where is the Emcure Walk-In Interview?
A: On 17 May 2026 (Sunday), 10 AM–3 PM at Hotel Suba Elite, Vadodara.
Q4: What documents should I carry?
A: Updated resume, CTC proof photocopies, valid ID proof, and highest qualification certificates.
Q5: What growth opportunities does Emcure offer?
A: Excellent learning, skill development, and fast career progression in a USFDA-compliant injectable manufacturing setup.


