Apotex is conducting a Walk-in Interview on 30th & 31st May 2026 in Mumbai for multiple Regulatory Affairs positions. Great opportunity for experienced professionals seeking pharma jobs, Regulatory Affairs jobs, and exciting pharmaceutical careers in India with a global Canadian company.
Contents
About the Company
Apotex is a proud Canadian-based global health company and one of the largest privately held pharmaceutical organizations in the world. With a strong presence in India through Apotex Research Private Limited (ARPL) in Mumbai and Bangalore, the company focuses on generics, biosimilars, innovative branded pharmaceuticals, and consumer health products.
Apotex is known for its commitment to innovation, highest standards of regulatory compliance, quality, and expanding access to affordable medicines across 45+ countries. The company offers a dynamic, collaborative environment with global exposure and excellent career growth opportunities.
Job Details
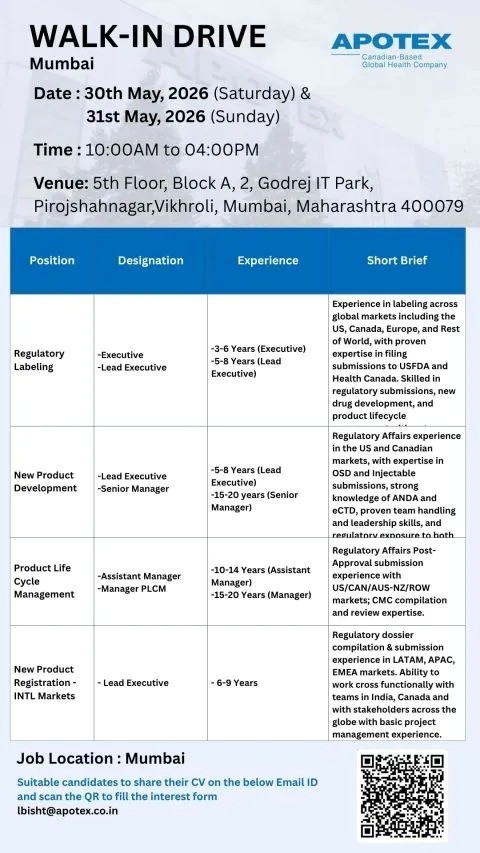
- Company Name: Apotex
- Experience: Varies by role (3-20 Years)
- Qualification: Relevant experience in Regulatory Affairs (specific qualifications not mentioned)
- Location: Mumbai, Maharashtra
- Work Type: Full-Time (On-site)
Job Description
Apotex is expanding its Regulatory Affairs team in Mumbai to support global product development, submissions, and lifecycle management. This walk-in drive offers multiple roles across key regulatory functions for OSD, Injectables, and international markets.
- Regulatory Labeling Department: Regulatory Affairs
- Role: Executive / Lead Executive
- Experience: 3-6 Years (Executive) | 5-8 Years (Lead Executive)
- Education/Qualification: Relevant Regulatory experience
- New Product Development Department: Regulatory Affairs
- Role: Lead Executive / Senior Manager
- Experience: 5-8 Years (Lead Executive) | 15-20 Years (Senior Manager)
- Education/Qualification: Relevant Regulatory experience
- Product Life Cycle Management (PLCM) Department: Regulatory Affairs
- Role: Assistant Manager / Manager
- Experience: 10-14 Years (Assistant Manager) | 15-20 Years (Manager)
- Education/Qualification: Relevant Regulatory experience
- New Product Registration – International Markets Department: Regulatory Affairs
- Role: Lead Executive
- Experience: 6-9 Years
- Education/Qualification: Relevant Regulatory experience
Skills/Qualifications
- Strong experience in USFDA, Health Canada, and international regulatory submissions
- Expertise in eCTD, ANDA, and CMC documentation
- Knowledge of labeling requirements across global markets
- Team handling and leadership skills for senior roles
- Cross-functional collaboration and project management abilities
- Excellent communication and problem-solving skills
Key Responsibilities
- Prepare and submit regulatory dossiers for new products
- Manage post-approval changes and lifecycle activities
- Review and update product labeling for global markets
- Compile CMC data and respond to regulatory queries
- Coordinate with global teams for submissions
- Ensure timely filings and compliance with regional requirements
Benefits/Perks
- Career growth in a leading global pharmaceutical company
- Exposure to international regulatory frameworks
- Learning opportunities with complex generics and biosimilars
- Collaborative and innovative work culture
- Competitive compensation and benefits
- Global networking and long-term career stability
How to Apply
Suitable candidates should share their CV at lbisht@apotex.co.in and scan the QR code to fill the interest form.
For more pharma jobs in Mumbai and across India, visit Pharma Recruiter. Apply promptly to secure your slot!
Walk-in Interview Details
- Date: 30th May 2026 (Saturday) & 31st May 2026 (Sunday)
- Time: 10:00 AM to 04:00 PM
- Venue: 5th Floor, Block A, 2, Godrej IT Park, Pirojshahnagar, Vikhroli, Mumbai, Maharashtra 400079
- Contact/Email: lbisht@apotex.co.in
Why You Should Join Apotex
Apotex offers a vibrant, purpose-driven culture where employees are empowered to make a real impact on global healthcare. With strong emphasis on innovation, regulatory excellence, and employee development, the company provides long-term career stability, recognition, and opportunities to work on cutting-edge projects alongside international teams in a supportive and inclusive environment.
FAQs
Q1: What experience is required for Apotex Regulatory Affairs roles?
A: Experience ranges from 3-6 years for Executive to 15-20 years for Senior Manager positions, depending on the role.
Q2: Which markets are covered in these regulatory jobs?
A: US, Canada, Europe, LATAM, APAC, EMEA, Australia-New Zealand, and ROW markets.
Q3: Is this a walk-in interview for all positions?
A: Yes, candidates can attend the walk-in on 30th & 31st May 2026 at the Mumbai venue.
Q4: What is the job location?
A: Mumbai (Vikhroli), Maharashtra.
Q5: How can I apply if I cannot attend the walk-in?
A: Email your CV to lbisht@apotex.co.in and fill the interest form via QR code.



