ADMAC Lifesciences Limited is hiring for multiple pharma jobs in Baddi and Panchkula. Openings available in QA, QC, Warehouse, Validation, Production, Regulatory Affairs, and more. Excellent opportunity for experienced professionals seeking pharmaceutical careers in India with attractive salary packages.
Contents
About the Company
ADMAC Lifesciences Limited is a growing pharmaceutical company based in Baddi, Himachal Pradesh. The company specializes in manufacturing high-quality injectable products and is committed to innovation, stringent regulatory compliance, and international quality standards.
With a modern manufacturing facility in the Baddi industrial area, ADMAC focuses on delivering safe and effective medicines while maintaining excellence in operations, quality assurance, and global regulatory requirements.
Job Details
- Company Name: ADMAC Lifesciences Limited
- Experience: Varies by role (0-2 years to 15+ years)
- Qualification: B.Pharm / M.Pharm / Relevant Science Graduate (preferred for most technical roles)
- Location: Baddi, Panchkula, Himachal Pradesh
- Work Type: On-site
Job Description
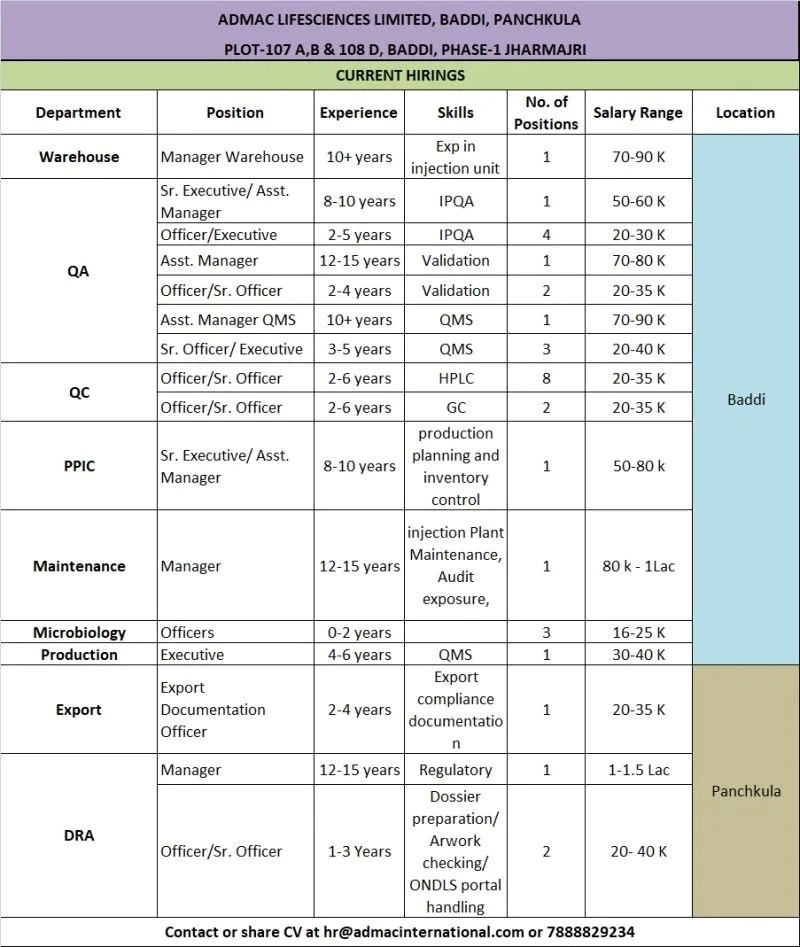
ADMAC Lifesciences Limited is actively hiring for multiple positions across various departments at its Baddi manufacturing plant. This is a great chance for professionals with injectable manufacturing experience to join a growing pharma company. Below are the key openings:
- Manager Warehouse Department: Warehouse
- Role: Warehouse Manager
- Experience: 10+ Years (Injection unit preferred)
- Education/Qualification: Relevant experience in Pharma Warehouse
- Asst. Manager / Sr. Executive – IPQA Department: IPQA (In-Process Quality Assurance)
- Role: IPQA
- Experience: 8-10 Years / 2-5 Years
- Education/Qualification: B.Pharm / M.Pharm
- Asst. Manager – Validation Department: Validation
- Role: Validation
- Experience: 12-15 Years
- Education/Qualification: Relevant Qualification
- Asst. Manager QMS Department: Quality Management System (QMS)
- Role: QMS
- Experience: 10+ Years
- Education/Qualification: B.Pharm / M.Pharm
- Officer/Sr. Officer – QC (HPLC / GC) Department: Quality Control
- Role: QC Analyst
- Experience: 2-6 Years
- Education/Qualification: B.Pharm / M.Sc
- Manager – Regulatory Affairs (DRA) Department: Regulatory Affairs
- Role: DRA Manager
- Experience: 12-15 Years (Dossier preparation)
- Education/Qualification: B.Pharm / M.Pharm
Other Openings: Production, PPIC, Maintenance, Microbiology, Export Documentation
Skills/Qualifications
- Experience in injectable manufacturing unit is highly preferred
- Strong knowledge of GMP, GLP, and regulatory guidelines
- Hands-on experience with HPLC, GC, Validation, and QMS
- Excellent documentation and compliance skills
- Ability to handle audits and regulatory inspections
- Good communication and team coordination skills
- Knowledge of export documentation and international filings
Key Responsibilities
- Manage warehouse operations and inventory control
- Perform IPQA and in-process quality checks
- Execute validation activities and protocols
- Handle QMS documentation and compliance
- Conduct HPLC/GC analysis in QC lab
- Prepare regulatory dossiers and manage filings
- Support production planning and maintenance activities
Benefits/Perks
- Attractive salary packages (16K to 1.5 Lac+)
- Career growth in a fast-growing injectable company
- Opportunities for skill development and learning
- Professional and supportive work culture
- Exposure to regulatory and international markets
- Stable employment with a quality-focused organization
How to Apply
Interested candidates can share their updated CV at hr@admacinternational.com or call 7888829234.
Mention the position you are applying for in the subject line.
Apply quickly for these pharma jobs in Baddi before the positions are filled.
For more QA jobs, QC jobs, production jobs, and regulatory openings across India, visit Pharma Recruiter.
Why You Should Join
ADMAC Lifesciences offers a dynamic work environment with strong emphasis on quality, compliance, and innovation. Employees get excellent opportunities for professional growth while working with modern injectable manufacturing facilities. The company values talent and provides long-term career stability in the highly regulated pharmaceutical sector.
FAQs
Q1. Which experience levels are eligible for ADMAC Lifesciences jobs?
A: Openings are available from freshers (0-2 years) to senior professionals (12-15 years) depending on the role.
Q2. Is injectable experience mandatory?
A: Yes, preference will be given to candidates with experience in injection units for most technical positions.
Q3. What is the location of these pharma jobs?
A: All positions are based in Baddi / Panchkula, Himachal Pradesh.
Q4. How can I apply for these openings?
A: Email your CV to hr@admacinternational.com or contact 7888829234.
Q5. What departments are hiring?
A: Warehouse, IPQA, QC, Validation, QMS, Production, Regulatory Affairs, Maintenance, and more.



