Exciting pharma jobs in Ahmedabad for Regulatory Affairs, Quality Control, and Production professionals. Orley Laboratories (WHO-GMP certified) is hiring for QC Head, QC Manager, RA Executive, and Production Manager roles at their Injectable Unit in Vatva. Apply now for these QC jobs and pharmaceutical careers in India.
Contents
About the Company
Orley Laboratories Pvt. Ltd. is a WHO-GMP certified pharmaceutical manufacturing company established in 1984 and headquartered in Ahmedabad, Gujarat. With state-of-the-art facilities in Vatva GIDC, the company specializes in critical care injectables and a wide range of formulations for domestic and international markets.
Known for its strong commitment to quality, regulatory compliance, and innovation, Orley Laboratories serves as a trusted contract manufacturer and global exporter. The company maintains ISO 9001:2015 certification along with multiple country-specific approvals, delivering high-quality pharmaceutical solutions while focusing on excellence in manufacturing and patient care.
Job Details
- Company Name: Orley Laboratories Pvt. Ltd.
- Experience: Varies by role (3–15+ years)
- Qualification: B.Sc / M.Sc / B.Pharm / M.Pharm
- Location: Vatva, Ahmedabad, Gujarat (Injectable Unit)
- Work Type: On-site
Job Description
Orley Laboratories Pvt. Ltd., a WHO-GMP certified injectable manufacturing company in Ahmedabad, is expanding its team with multiple senior openings in Regulatory Affairs, Quality Control, and Production/Outsourcing. These roles offer excellent opportunities for experienced professionals seeking growth in the Indian pharmaceutical industry, particularly those with parenteral/injectable experience.
- RA Executive Department: Regulatory Affairs (RA)
- Experience: Minimum 3 years in Regulatory Affairs (formulations)
- Education/Qualification: B.Pharm / M.Pharm
- Vacancies: 2
- QC Manager Department: Quality Control (QC)
- Experience: Minimum 5 years in Quality Control (pharma)
- Education/Qualification: B.Sc / M.Sc / B.Pharm / M.Pharm
- Vacancies: 1
- QC Head Department: Quality Control (QC)
- Experience: Minimum 15 years in Quality Control
- Education/Qualification: B.Sc / M.Sc / B.Pharm / M.Pharm
- Vacancies: 1
- Production Manager (Outsourcing and Quality Assurance) Department: Production / Outsourcing & QA
- Experience: Senior level with extensive relevant experience
- Education/Qualification: B.Sc / M.Sc / B.Pharm / M.Pharm
- Vacancies: 1
Skills/Qualifications
- Experience in dossier preparation (CTD/ACTD) and regulatory submissions for RA role
- Hands-on experience with HPLC, method validation, stability studies, and OOS/OOT investigations for QC positions
- Strong team leadership and audit-handling skills, especially for QC Head
- GMP compliance knowledge and documentation expertise across all roles
- Willingness to travel extensively with own vehicle for Production Manager role
- Parenteral/injectable manufacturing or analytical experience preferred
- Excellent coordination and communication abilities
Key Responsibilities
- Prepare and submit dossiers (CTD/ACTD) for domestic and export markets
- Manage lab operations, stability studies, and investigations (QC Manager)
- Lead overall QC function, audits, and regulatory inspections (QC Head)
- Monitor third-party manufacturing sites for GMP compliance
- Handle product registrations, variations, and authority coordination
- Identify deviations and implement corrective actions
- Ensure documentation control and quality system adherence
Benefits/Perks
- Career growth in a WHO-GMP certified organization
- Opportunities to work on critical care injectables and global projects
- Professional development in regulatory and quality domains
- Collaborative work culture with focus on excellence
- Competitive salary and long-term stability
How to Apply
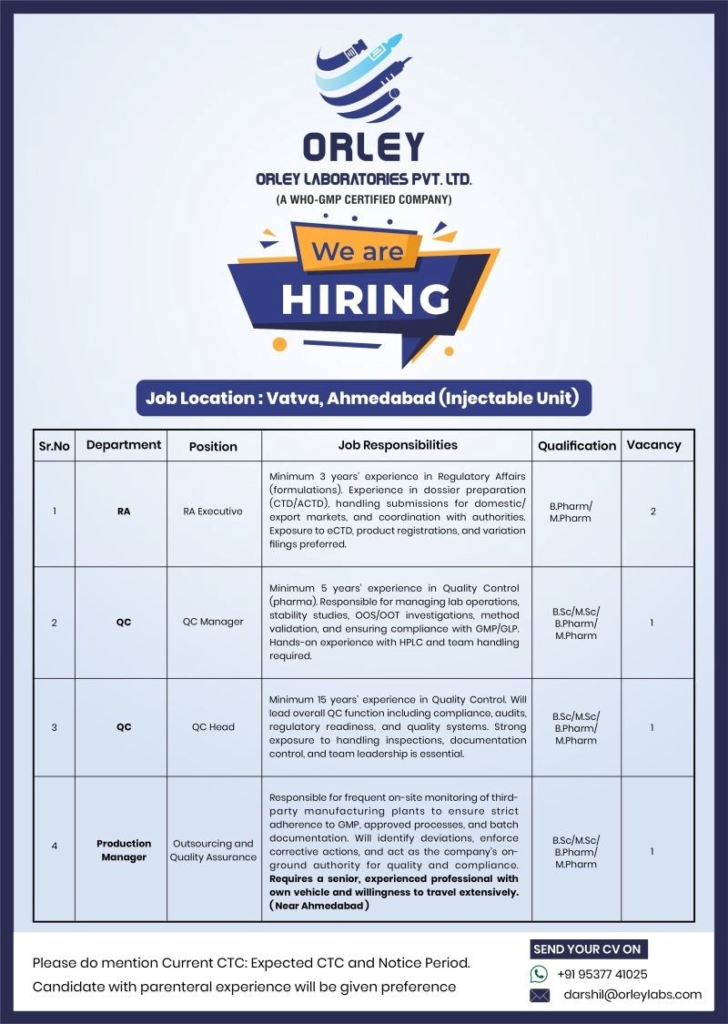
Interested candidates should send their updated CV mentioning Current CTC, Expected CTC, and Notice Period to:
Email: darshil@orleylabs.com Phone/WhatsApp: +91 95377 41025
Candidates with parenteral/injectable experience will be given preference.
Apply immediately for these pharma jobs in Ahmedabad. For more QC jobs, QA jobs, production jobs, and pharmaceutical careers across India, visit Pharma Recruiter.
Why You Should Join
Orley Laboratories offers a stable, growth-oriented environment backed by decades of experience in pharmaceutical manufacturing. Join a company recognized for quality excellence, global reach, and strong regulatory compliance. You will gain valuable exposure to injectable manufacturing, international standards, and cross-functional leadership roles while contributing to essential healthcare solutions.
FAQs
Q1: What experience is required for the QC Head position at Orley Laboratories?
A: Minimum 15 years in Quality Control with leadership experience in audits and regulatory compliance.
Q2: Are these on-site jobs in Ahmedabad?
A: Yes, all positions are on-site at the Injectable Unit in Vatva, Ahmedabad.
Q3: What should I include in my application for these pharma jobs?
A: Mention Current CTC, Expected CTC, Notice Period, and highlight any parenteral experience.
Q4: Is injectable experience mandatory?
A: It is not mandatory but candidates with parenteral/injectable experience will be given strong preference.
Q5: How many vacancies are available?
A: Total 5 vacancies across RA Executive (2), QC Manager (1), QC Head (1), and Production Manager (1).



